
They have a high melting and boiling point. It includes the elements lanthanide and actinide. E.g., Ag The valance electron lies in the d sub-shells. 
It includes the transition element, which lies in between the s and p block elements. The elements in which the last electrons enter any one of the five d-orbitals of their respective penultimate shell are d-block elements. The completely filled p-orbital are noble gases. They have 1-6 valence electrons in the outermost shells. It includes metals, metalloids, nonmetals, and inert gases. The elements in which the last electron enters into any of the p-orbitals of the outermost shell are called p-block elements. Most elements of this block impart characteristics of clour to the flame. They are good reducing agents since they are highly electropositive and form positive ions by losing one or two electrons from their outermost shells. It includes alkali and alkali earth metals. They are very soft metals with low melting and boiling points. Metallic character, chemical reactivity of metals increases but not metallic character and chemical reactivity decreases as we move down the group.Įach shell consists of a number of sub-shells in which electrons are distributed.Ionization increases as we move down the group.Atomic size increases as we move down as one new shell is added.Elements in the group have same valency except for the group 0, which do not take part in the chemical reaction.ar.Atoms of the element in the group have the same number of the electron in the outermost shell.Groups are the vertical columns of elements in periodic table.Electronegativity and metallic character increases as we move from left to right.Ionization energy and electro positivity decreases as we move from left to right in periodic table.Atomic size of an atom decreases from left to right due to increase in nuclear charge as extra electron is added.The valency of the electron increases from 1 to 4 then decreases to 0.The number of electrons in the valance shell increases as we move from the left to right. Lanthanides(4f-series) and Actinides(5f-series) are separated rows at the bottom of periodic table, these elements are called Inner transitional elements. Inert gasesĮlements of zero group or (18) column are called inert gases.Įlements constituting subgroup B (IB, IIB, IIIB, IVB, VB, VIB, VIIB) and VIII are called transition elements. Two groups on the left (IA &IIA) and five groups on left (IIA, IVA, VA, VIA, VIIA) of Subgroup A are representative elements. Bohr (1920) constructed a periodic table known as the long form periodic table.ĭepending on the number of incomplete shells in the atom, the elements in the periodic table can be classified into four different types of elements. /periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
Periods are the horizontal rows of elements in the periodic table. Elements have been classified into 4 different blocks.The controversy of Hydrogen was clarified.Isotopes of the same element can be placed within the same group due to the same atomic number.Some elements, such as Argon and Potassium, Cobalt and Nickel, were reorganized based on their atomic number.The features of Modern Periodic Table are: So it is superior to Mendeleev’s periodic table. Modern periodic table rules state that “The physical and chemical properties of elements are a periodic function of their atomic number.”Įlements are grouped into four different blocks, in ascending order of atomic number, and thus the positions of alkali metals, alkali earth metals, halogens, inert gases, and other elements are separated in the modern Periodic Table. 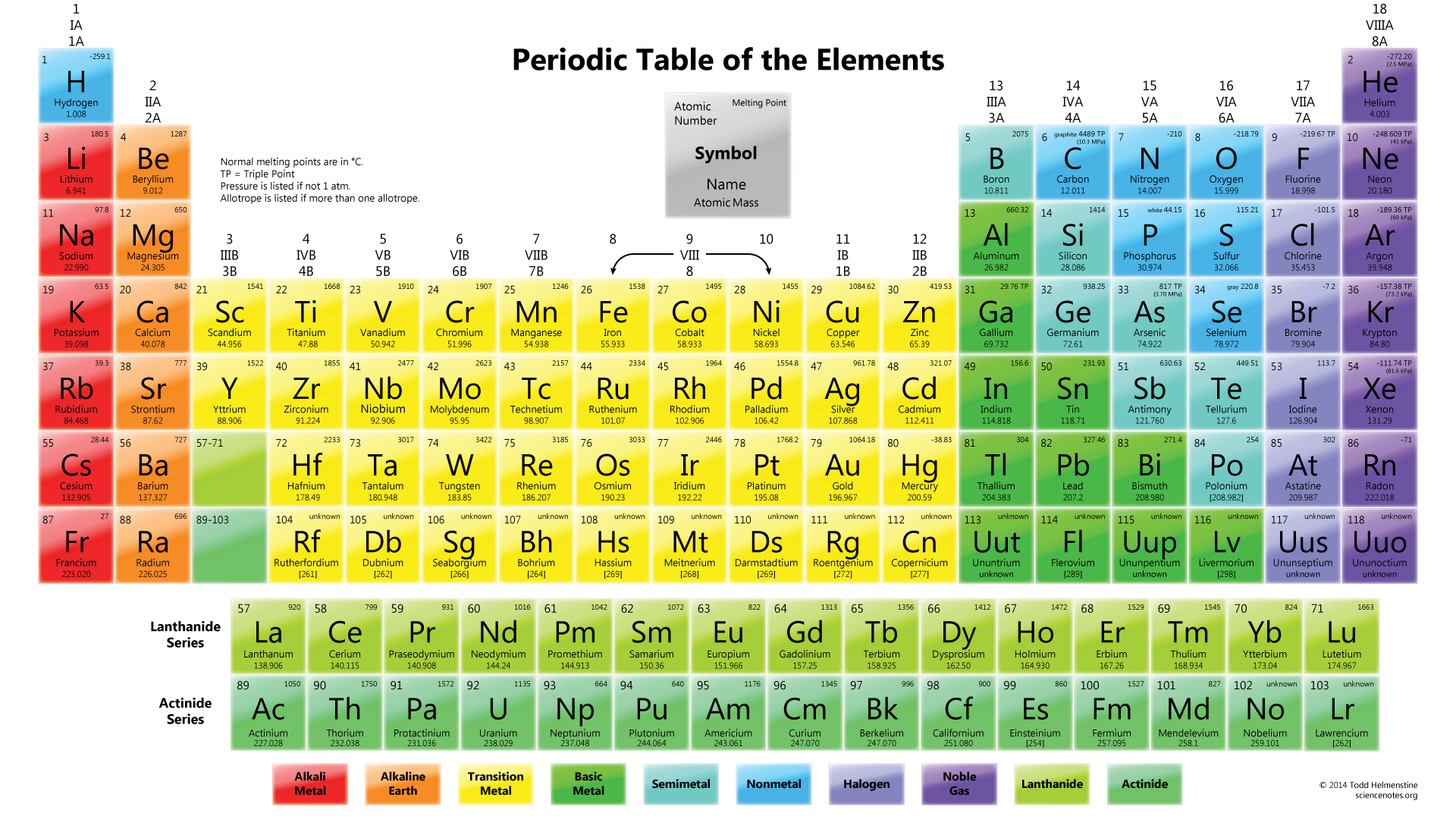
In 1912, Moseley, an English physicist, observed that the physical and chemical properties of the elements are determined by atomic number rather than atomic weight.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
